It was determined, that such hormone as testosterone stimulates the general growth of the bone matrix and contributes to the retention of calcium in organism. The bone matrix growth is correlated with the anabolic activity of testosterone, and the formation of calcium deposits is a secondary effect, caused by the growth of bone matrix. The metabolic activity of testosterone is intermediated by the androgenic receptor in the cell cytoplasm. Within the cell cytoplasm the testosterone is binding to cell’s receptor (protein). After the complex was formed, the testosterone becomes activated and then is transported to the cell nucleus. The binding of the complex to chromosomal proteins starts the formation process of messenger ribonucleic acid (mRNA), necessary for synthesis of proteins, which are responsible for testosterone activity. Receptor can interact with testosterone and some other androgens. Within osseous tissue cells and skeletal muscle fibers, testosterone possesses the higher sensibility to the receptor than other androgens. Within other cells the highest sensibility to the receptor showed 5a-dihydrotestosterone. In this case, after testosterone was within the cell, it is to be transformed into 5a-dihydrotestosterone. In the skeletal muscles and osseous tissue this transformation of testosterone into 5a-dihydrotestosterone is impossible. The genetic information, contained in mRNA, is transferred within the process of cell’s proteins synthesis in ribosome.
The state of the cell’s receptors is regulated by increase or decrease of binding locations and binding sensibility changes. Thus, the testosterone effect on metabolic processes depends from its source, as well as from stimulation or inhibition of specific cell’s receptor function; hence, the increasing of metabolic hormonal influence may be conditioned not only by high concentration of hormone in the intercellular fluid, but also by as a result of increase of binding locations and/or increased receptor’s sensibility to hormone. When receptor’s function is reduced, even the high hormone level will be incapable to influence effectively on the metabolism. Therefore it is incorrect to equal the concentration of hormone in blood and the degree of its effect on metabolism.
Effect of testosterone among women may be increased dependent of estrogens output (Danhaive, Rousseau, cit. [20]). The increased sensibility to testosterone is believed to be due to the stimulation of the function of androgenic receptors.
Basing on these facts, the authors suppose:
- Testosterone has an effect on the bone matrix growth;
- Keeping of appropriate testosterone level is important for woman’s organism;
- Testosterone effect on woman’s organism may be strengthened by estrogens [2,3].
The main androgens in the blood serum of women with normal menstrual cycle are testosterone and dihydrotestosterone. Dihydroepiandrosterone sulfate (DHAE-sulfate), dihydroepiandrosterone (DHAE), and androstenedione are estimated as prohormones since their androgenic properties become effective only after their conversion into testosterone. Organism of an average healthy woman of reproductive age secretes ca. 300 μg of testosterone per day (i.e. 5% from the average per day testosterone output among men). With ageing the amount of androgens in female organism is being significantly reduced. Thus, the average levels of bioavailable and free testosterone, androstendione and dihydroepiandrosterone (DHAE) among the 45 years old women amount 50%, among 60 years old – ca. 30%, among 70 years old – 10% in comparison with the average level for 20 years old women (100%) [12]. As was reported in some researches, the endogenous androgens showed the favorable effect on the bone mineral density among the women after menopause [10]. On the base of abovementioned facts, S.Kalinchenko et al. (2008) supposed, that investigation of the role of androgens in bone metabolism is completely reasonable and necessary for further treatment of osteoporosis and osteopenia, especially among women.
At present the world practice has a great experience of hormone replacement therapy (HRT) based on administration of estrogens. However there are more an more data, which confirm, that the effective treatment of osteoporosis is often impossible without correcting the age-dependent androgenic deficiency (hypogonadism). Furthermore, the wide use of hormone (estrogen) replacement therapy, especially among aged women, may be limited because of series of contraindications, such as various diseases of liver, risk of thrombus developing, thrombophlebitis, risk of endometrial hyperplasia and hyperplasia of the breast. During prolonged (10-15 years) administration of estrogens the risk of cancer increases in 30-50% [10].
Considering all above mentioned, it may be concluded that the exploring of further new methods of effective treatment of senile osteoporosis is extremely actual. As since administration of natural hormones (estrogens and androgens) has several disadvantages, it was recommended to use herbal hormones. So, in USA was developed a substrate (Citracal plus Vitamin D plus Genistein), intended for synthesis of organism’s own hormones, containing calcium citrate and genistein (isoflavone, extracted from soybeans). We (Parapharm Ltd.Co. and Penza institute of physicians’ advanced training) have developed new technologies in treatment for osteoporosis with addition of such bee keeping articles as drone brood as donator of sex hormones (Patent №2412616) [14,15,16,17].
One capsule of our new remedy «Osteomed-forte» contains vitamin D3 – 500 IU per day, drone brood – 100 mg, calcium citrate – 500 mg. The necessity of the adding of drone brood was demanded by its properties as donator of sex hormones, such as estradiol, progesterone and testosterone, which provide stimulating effect on the bone mineral density. Drone brood is used for treatment of sexual disfunctions, caused by androgenic deficiency, and climax [18].
By the clinical researches it was determined that senile osteoporosis induce the formation of cavities in cancellous (trabecular) bones (trabeculation) as a result of increased resorption of bones’ trabeculae. However, there is no published researches concerning the problem of closing of cavities, caused by senile osteoporosis. Penza institute of physicians’ advanced training is focused now on the investigation of various possible osteoprotectors, which could be capable to close such bone cavities.
Research aim.
Investigation of «Osteomed – forte» activity for the treatment of postmenopausal and senile osteoporosis among women with androgenic deficiency.
Materials and methods.
In the period from 2009 to March 2012 The Osteoporosis research center of Penza institute of physicians’ advanced training had examined 10 patients – women from 49 to 85 years old (an average age consisted 65,5 ± 3,7 years). The patients were selected for examination according to following criteria: women with postmenopausal and senile osteoporosis, hormonally and clinically confirmed androgenic deficiency, bone mineral density less than – 2,5 of standard deviation (SO) and presence of cavities in trabecular bones. Women with secondary osteoporosis and various systemic and endocrine diseases, oncologic pathologies, as well as who was been treated with estrogen replacement therapy (ERT) or glucocorticoids were excluded from this investigation. All patients had signed a written agreement on their participation in the clinical testing of the «Osteomed forte» compound.
The diagnostics of osteoporosis were conducted on the base of complaints and physical examinations of patients with use of medical, radiologic and clinical biochemistry tests. For the determining of bone mineral density by X-ray absorptiometry method was been used device «Osteometr – DTX – 100». It is the only type of device that can simultaneously detect bone mineral density, presence of cavities in bones and their dynamics. The examination of patients included collection of medical histories, general examination, medical tests (incl. radiologic tests and clinical chemistry), as well as hormone investigations to determine amounts of bioavailable testosterone, sex hormone-binding globulin (SHBG), thyroid-stimulating hormone (to exclude patients with hypothyroidism). Hormone investigation was performed using immuno chemiluminescence method and «Immulite 2000» device. Seriousness of osteoporosis was evaluated according to the Classification of World Health Organization (WHO).
As criteria for evaluation of activity of «Osteomed forte» for the treatment of age-depending osteoporosis were taken the results of clinical chemistry, radiologic and medical tests, which were intended to examine the capacity of closing or diminishing of cavities in trabecular bones.
Depending of the method of treatment of osteoporosis all the tested women were divided into two groups, correlated by age and seriousness of disease. The first group included 37 women, to which was administered «Osteomed forte», taken orally, 1 capsule two times per day at morning and before sleep; the treatment included 3-month course of treatment three times in the year. (One capsule of «Osteomed forte» contained 500 mg of calcium citrate, 100 mg of drone brood, 500 IU of vitamin D3). The second group (control) included 33 women, to which was taken «Calcium D3 Nicomed», containing in one capsule 0,5 g of Calcium, using the same scheme of administering.
All patients were examined before the administration of remedies and after each three months of therapy up to the end of the whole course of treatment in order to determine bone mineral density and dynamics of development of bone cavities using X-ray absorptiometry method.
Statistical assay of the received data was performed using the program packet StatSoft, Windows XP. Quantitative values are described taking in account the average and standard deviations. Received data are presented in the format M±m, where М is an arithmetic mean, and m – error of mean. Distinctions were considered as statistically significant when significance level is р<0,05 [19].
Results and discussion.
After the end of treatment course 75% of patients from the first group of women, treated with «Osteomed forte», showed the more visible therapeutic effect: 50% of patients – diminishing of bone cavities, 25% – closing of bone cavities. In the second group of tested, treated with «Calcium D3 Nicomed», 60% of patients had no favorable effects and the closing of bone cavities occurred 5 times more seldom than in first group. The better results of treatment with «Osteomed forte» can be explain by the presence of drone hormones, serving as a substrate for synthesis of necessary hormones in patient’s own organism.
The analysis of hormone values showed that concentrations of available testosterone among women from two tested groups before treatment were: in the first group – 1,1±0,4 nmol/l, in the second group – 1,2±0,5 nmol/l (р>0,05) at reference ranges for present method 1,7-3,4 nmol/l. Concentrations of sex hormone-binding globulin in the both groups before treatment were 64,3 ± 2,6 nmol/l, 62,8 ± 2,9 nmol/l (р>0,05), respectively. Nine months later, after the treatment with «Osteomed forte» was remarked improvement of state among 29 from 37 patients. Medical tests showed increase of available testosterone concentration in blood serum from 1,1±0,4 nmol/l (before treatment) to 2,5±0,6 nmol/l (after treatment), as well as increase of the sex hormone-binding globulin from 64,3±2,6 nmol/l to 115,0±5,9 nmol/l (р<0,05), that exceeded the upper limit of reference ranges for blood tests 16–108 nmol/l. Patients from the second group, treated with «Calcium D3 Nicomed», showed no positive changes neither in testosterone level nor increase of sex hormone-binding globulin. The results of investigation of bone mineral density in the both groups after 9 months of treatment are showed in Figure 1.
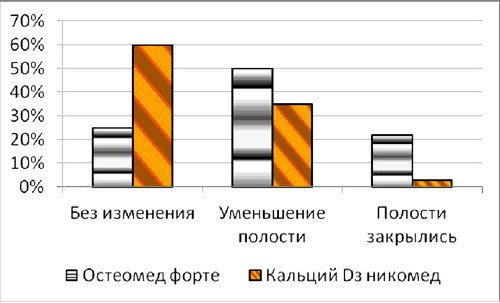
Fig. 1. Bone mineral density in the both groups after 9 months of treatment.
As it was shown in the Figure 1, among the first group, treated with «Osteomed forte», only 24% of patients reached no positive changes, whereas in the second group, treated with «Calcium D3 Nicomed», this rate was 60%. Diminishing of bone cavities’ size in the first group, treated with «Osteomed forte», was 50%, whereas in the second group, treated with «Calcium D3 Nicomed», this rate was 36%. The rate of closing of bone cavities in first group («Osteomed forte») was 5 times higher than in the second group («Calcium D3 Nicomed»). Figures 2-7 demonstrate closing of bone cavities with an example of three women from the first group, treated with «Osteomed forte». The osteometry, performed before treatment detected bone cavities (Fig. 2, 4, 6 – before treatment). After 10 months of treatment with «Osteomed forte» cavities have closed (Fig. 3, 5, 7).




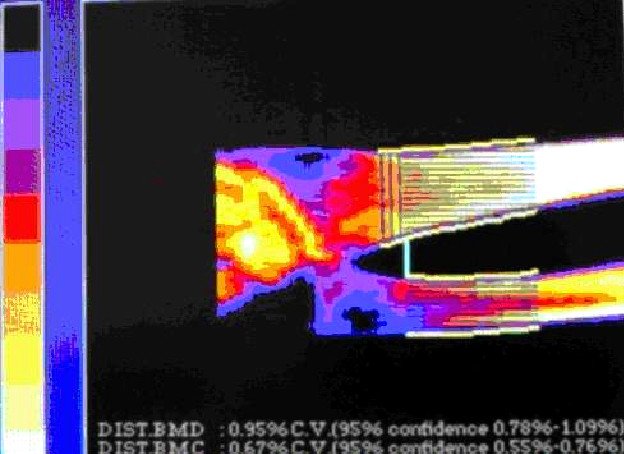 Fig.2. Patient S., 52 years old. Bone cavities before treatment.
Fig.2. Patient S., 52 years old. Bone cavities before treatment.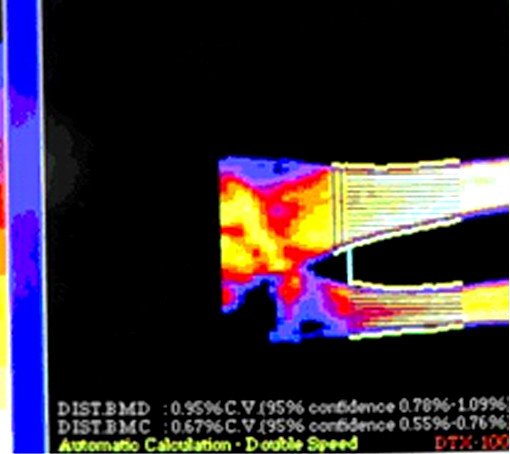
 Fig.6. Patient E., 72 years old. Bone cavities before treatment.
Fig.6. Patient E., 72 years old. Bone cavities before treatment.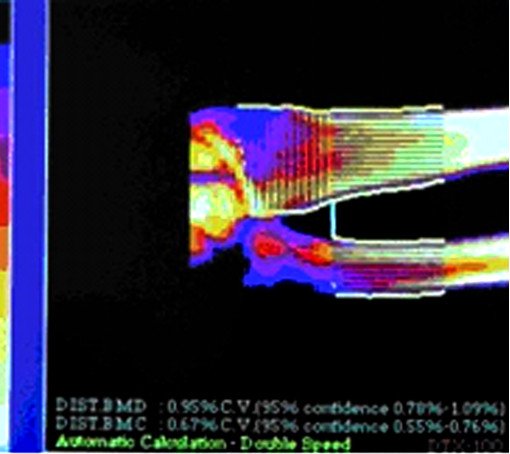 Fig. 3. Patient S. Bone cavities have closed after 10 months treatment.
Fig. 3. Patient S. Bone cavities have closed after 10 months treatment. Fig. 5. Patient K. after 10 months treatment. Cavities have closed
Fig. 5. Patient K. after 10 months treatment. Cavities have closed Fig.7. Patient E. after 10 months treatment. Cavities have closed.
Fig.7. Patient E. after 10 months treatment. Cavities have closed.